iStock
How chronic stress leads to hair loss
Harvard study IDs mechanism that regenerates hair follicle stem cells
Harvard University researchers have identified the biological mechanism by which chronic stress impairs hair follicle stem cells, confirming long-standing observations that stress might lead to hair loss.
In a mouse study published in the journal Nature, the researchers found that a major stress hormone puts hair follicle stem cells into an extended resting phase, without regenerating the follicle or the hair. The researchers identified the specific cell type and molecule responsible for relaying the stress signal to the stem cells, and showed that this pathway can be potentially targeted to restore hair growth.
“My lab is interested in understanding how stress affects stem cell biology and tissue biology, spurred in part by the fact that everyone has a story to share about what happens to their skin and hair when they are stressed. I realized that as a skin stem cell biologist, I could not provide a satisfying answer regarding if stress indeed has an impact — and more importantly, if yes, what are the mechanisms,” said Ya-Chieh Hsu, the Alvin and Esta Star Associate Professor of Stem Cell and Regenerative Biology at Harvard and senior author of the study. “The skin offers a tractable and accessible system to study this important problem in depth, and in this work, we found that stress does actually delay stem-cell activation and fundamentally changes how frequently hair follicle stem cells regenerate tissues.”
The hair follicle is one of the few mammalian tissues that can undergo rounds of regeneration throughout life, and has become a paradigm that informs much of our fundamental understanding of mammalian stem cell biology. The hair follicle naturally cycles between growth and rest, a process fueled by hair follicle stem cells. During the growth phase, hair follicle stem cells become activated to regenerate the follicle and hair, and hairs grow longer each day. During the resting phase, the stem cells are quiescent and hairs shed more easily. Hair loss can occur if the hairs shed and the stem cells remain quiescent without regenerating new tissue.
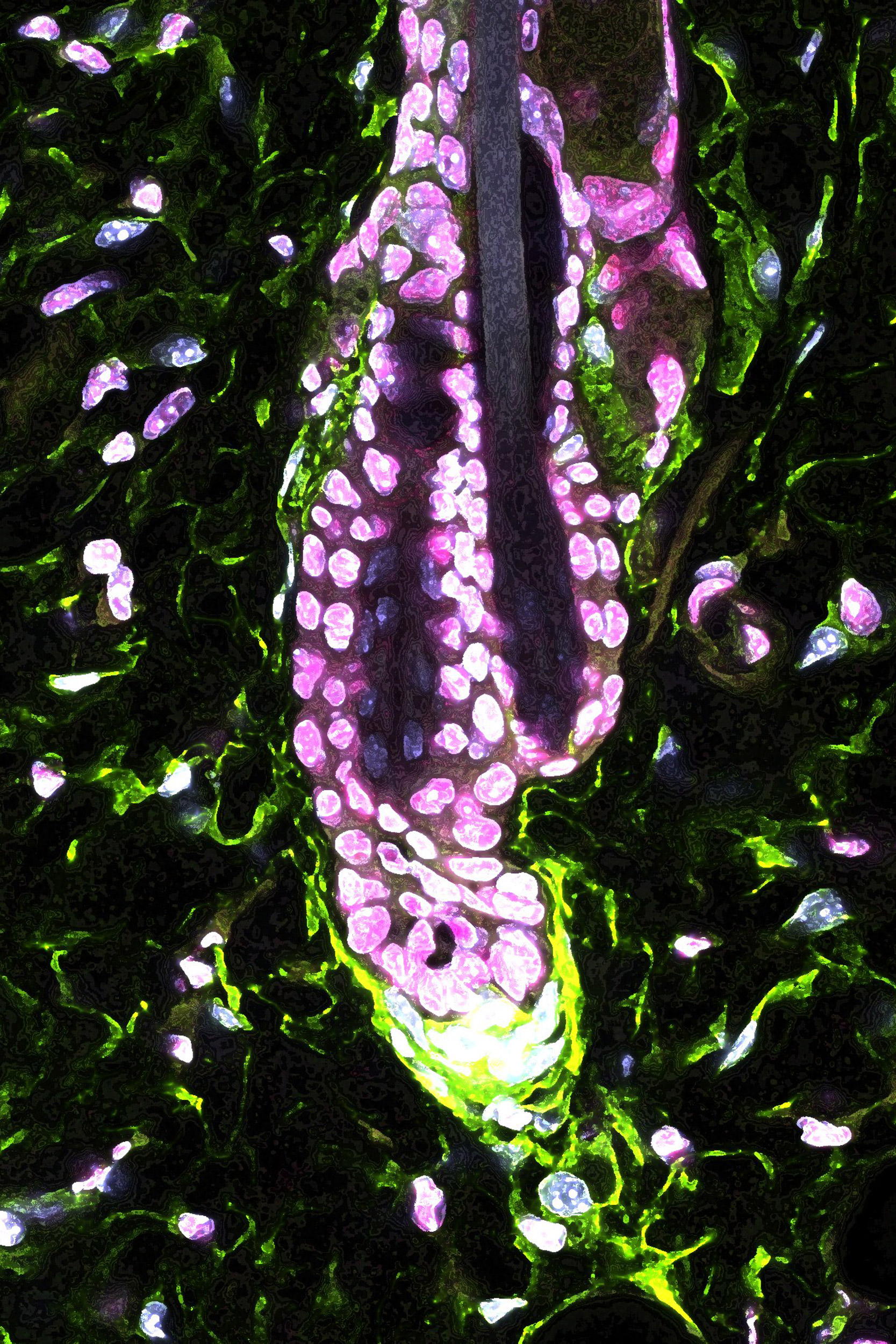
The researchers studied a mouse model of chronic stress and found that hair follicle stem cells stayed in a resting phase for a very long time without regenerating tissues. A major stress hormone produced by the adrenal glands, corticosterone, was upregulated by chronic stress; giving mice corticosterone reproduced the stress effect on the stem cells. The equivalent hormone in humans is cortisol, which is also upregulated under stress and is often referred to as the “stress hormone.”
“This result suggests that elevated stress hormones indeed have a negative effect on hair follicle stem cells,” Hsu said. “But the real surprise came when we took out the source of the stress hormones.”
Under normal conditions, hair follicle regeneration slows over time — the resting phase becomes longer as the animals age. But when the researchers removed the stress hormones, the stem cells’ resting phase became extremely short and the mice constantly entered the growth phase to regenerate hair follicles throughout their life, even when they were old.
“So even the baseline level of stress hormone that’s normally circulating in the body is an important regulator of the resting phase. Stress essentially just elevates this preexisting ‘adrenal gland–hair follicle axis,’ making it even more difficult for hair follicle stem cells to enter the growth phase to regenerate new hair follicles,” Hsu said.
After establishing the link between the stress hormone and hair follicle stem cell activity, the researchers looked for the biological mechanism underlying the connection.
“Under both normal and stress conditions, adding Gas6 was sufficient to activate hair follicle stem cells that were in the resting phase and to promote hair growth.”
Sekyu Choi, lead author of the study
“We first asked whether the stress hormone was regulating the stem cells directly and checked by taking out the receptor for corticosterone, but this turned out to be wrong. Instead, we found that the stress hormone actually acts on a cluster of dermal cells underneath the hair follicle, known as the dermal papilla,” said Sekyu Choi, the lead author of the study.
Dermal papilla is known to be critical for activating hair follicle stem cells, but none of the previously identified factors secreted from dermal papilla changed when stress hormone levels were altered. Rather, the stress hormone prevented dermal papilla cells from secreting Gas6, a molecule that the researchers showed can activate the hair follicle stem cells.
“Under both normal and stress conditions, adding Gas6 was sufficient to activate hair follicle stem cells that were in the resting phase and to promote hair growth,” Choi said. “In the future, the Gas6 pathway could be exploited for its potential in activating stem cells to promote hair growth. It will also be very interesting to explore if other stress-related tissue changes are related to the stress hormone’s impact on regulating Gas6.”
More like this
These initial findings in mice need to be further studied before they can be safely applied to humans. Harvard’s Office of Technology Development has protected the intellectual property relating to this work and is exploring opportunities for collaboration on its further development and eventual commercialization.
Last year, Hsu’s group discovered how stress affects another type of stem cell in the hair follicle: the melanocyte stem cells that regenerate hair pigment. The researchers found that stress activates the sympathetic nervous system and depletes melanocyte stem cells, leading to premature hair graying. With the new study, the two findings together demonstrate that although stress has detrimental impacts on both hair follicle stem cells and melanocyte stem cells, the mechanisms are different. Stress depletes melanocyte stem cells directly via nerve-derived signals, while stress prevents hair follicle stem cells from making new hairs indirectly via an adrenal-gland-derived stress hormone’s impact on the niche. Because hair follicle stem cells are not depleted, it might be possible to reactivate stem cells under stress with mechanisms such as the Gas6 pathway.
Beyond the potential application of the Gas6 pathway in promoting hair growth, the study’s results have broader implications for stem cell biology.
“When looking for factors that control stem cell behaviors, normally we would look locally in the skin. While there are important local factors, our findings suggest that the major switch for hair follicle stem cell activity is actually far away in the adrenal gland and it works by changing the threshold required for stem cell activation,” Hsu said.
“You can have systemic control of stem cell behavior located in a different organ that plays a really important role, and we are learning more and more examples of these ‘cross-organ interactions.’ Tissue biology is interconnected with body physiology. We still have so much to learn in this area, but we are constantly reminded by our findings that in order to understand stem cells in the skin, we often need to think beyond the skin.”
This work was supported in part by the New York Stem Cell Foundation, Smith Family Foundation Odyssey Award, Pew Charitable Trusts, Harvard Stem Cell Institute, Harvard HMS Dean’s Award, American Cancer Society, James and Audrey Foster MGH Research Scholar Award, NARSAD Young Investigator Award, MGH ECOR Fund, New York State Department of Health, and the National Institutes of Health.